Publications
Highlighted Publications
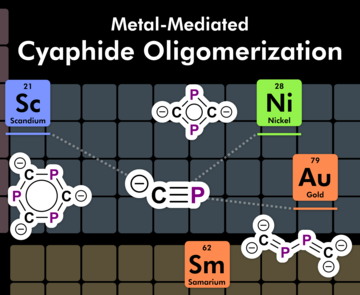
Metal-Mediated Oligomerization Reactions of the Cyaphide Anion
The cyaphide anion, CP−, is shown to undergo three distinct oligomerization reactions in the coordination sphere of metals. Reductive coupling of Au(IDipp)(CP) was found to afford a tetrametallic complex containing a C2P24– fragment. By contrast, non-reductive dimerization of Ni(SIDipp)(Cp)(CP) gives rise to an asymmetric bimetallic complex containing a C2P22– moiety. Spontaneous trimerization of Sc(Cp*)2(CP) results in the formation of a trimetallic complex containing a C3P33– fragment. These transformations show that while cyaphido transition metal complexes can be readily accessed using metathesis reactions, many such species are unstable to further oligomerization processes.
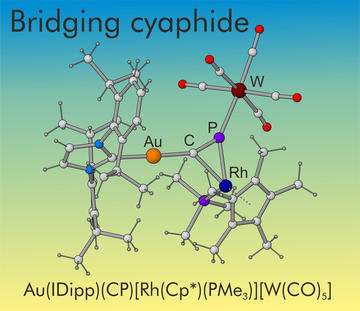
Revealing the Role of the Cyaphide Ion as a Bridging Ligand in Heterometallic Complexes
Hetero bi- and tri-metallic transition metal complexes featuring bridging cyaphide ions (C≡P−) are reported. These were synthesized from reactions of Au(IDipp)(CP) with electron-rich, nucleophilic transition metal reagents. These studies suggest that the reactivity of Au(IDipp)(CP) is largely LUMO-driven and that it behaves as a metallo-phosphaalkyne.
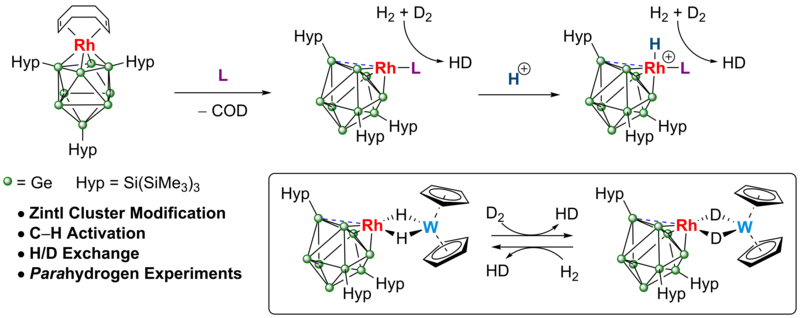
Zintl cluster supported low coordinate Rh(I) centers for catalytic H/D exchange between H2 and D2
We describe that modification of the catalytically active [Rh(COD){Ge9(Hyp)3}] by ligand substitution with phosphines or carbenes provides access to hypercloso clusters of form [Rh(L){Ge9(Hyp)3}], which feature coordinatively unsaturated rhodium centers and are catalytically active in H–H and C–H bond activation processes. To offer experimental insight into the mechanism of catalysis, isotopic labelling and parahydrogen measurements were also conducted.
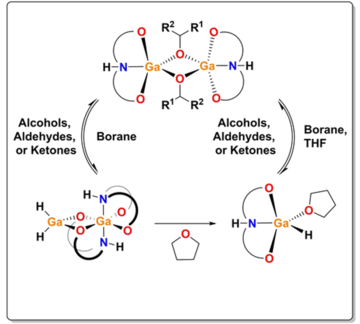
Pincer-Supported Gallium Complexes for the Catalytic Hydroboration of Aldehydes, Ketones and Carbon Dioxide
We describe the preparation of two novel pincer ligand supported gallium hydride complexes. The reactivity of these complexes towards aldehydes and ketones reveal the formation of alkoxide-bridged dimers through 1,2-hydrogallation reactions. The regeneration of the hydrides, through Ga−O/B−H metathesis with HBpin and 9-BBN, allowed us to demonstrate the catalytic reduction of carbonyl substrates with low catalyst loadings at room temperature.
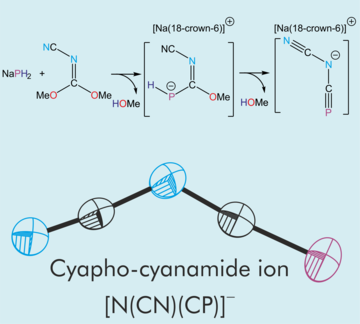
Synthesis, structure and reactivity of a cyapho-cyanamide salt
We describe a facile synthesis of the cyapho-cyanamide salt. Further reactivity studies of this anion reveal that functionalization at the phosphorus atom is viable to yield a new family of cyanide-functionalised phosphorus heterocycles.
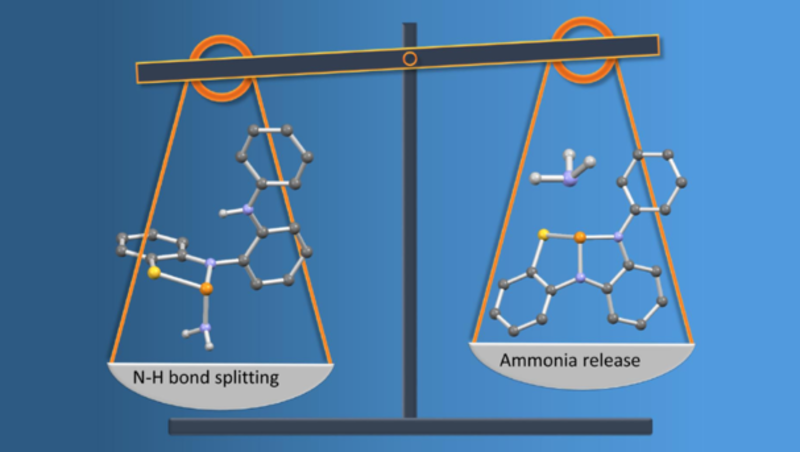
Thermoneutral N–H bond activation of ammonia by a geometrically constrained phosphine.
A geometrically constrained phosphine bearing a tridentate NNS pincer ligand is reported. The effect of the geometric constraint on the electronic structure was probed by theoretical calculations and derivatization reactions.
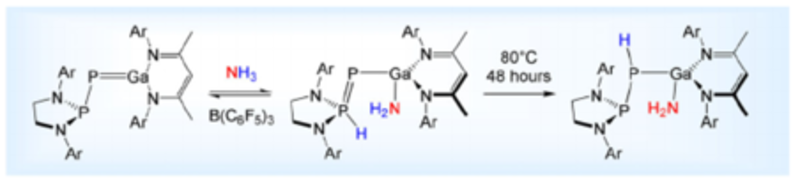
Contrasting E−H Bond Activation Pathways of a Phosphanyl-Phosphagallene
The reactivity of a phosphanyl-phosphagallene toward E–H bonds is reported. A frustrated Lewis pair type of mechanism operates at room temperature. At high temperatures, addition across the Ga=P bond is accessed by silanes or alkyl amines.
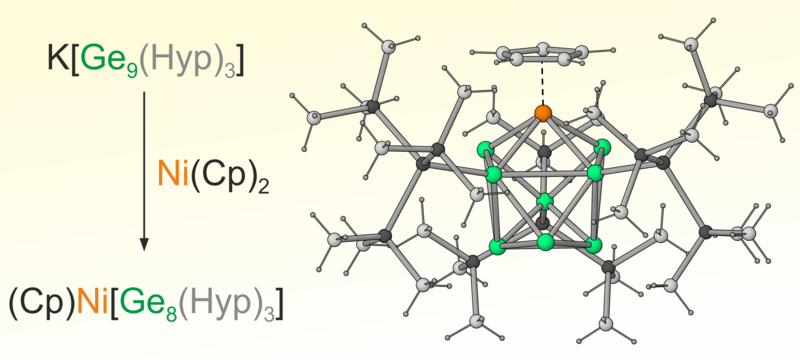
Cluster expansion and vertex substitution pathways in nickel germanide Zintl clusters
The reactivity of the Zintl cluster K[Ge9(Hyp)3] towards the nickel reagents Ni(COD)2 and Ni(Cp)2 is studied. This results in two remarkably different nickel germanide clusters via either cluster expansion or vertex substitution. The products were analysed by cyclic voltammetry and showed reversible redox behaviour for both clusters.
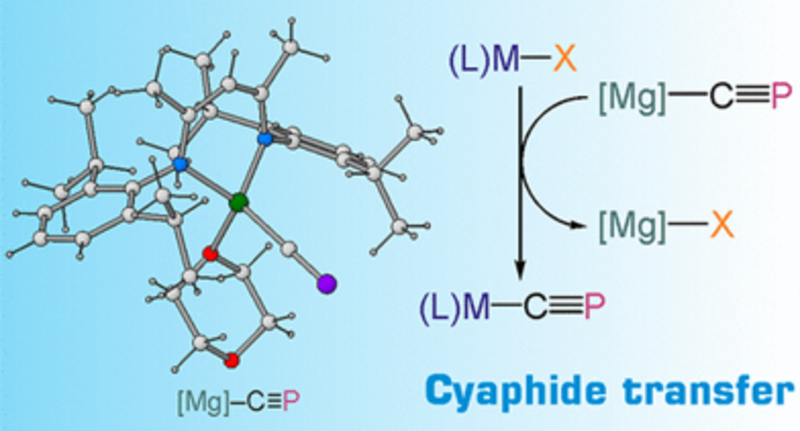
A Cyaphide Transfer Reagent
The cyaphide ion is a heavy analogue of the well-known cyanide ion. We have reported a magnesium cyaphide complex which can be used as a cyaphide transfer reagent. By analogy to Grignard reagents, it can be used to prepare metal cyaphide complexes by salt metathesis.